When added to an antidepressant,
REXULTI® (brexpiprazole) 2 mg/day was superior in reducing mean MADRS total score vs antidepressant + placebo
Mean Change from Baseline in MADRS Total Score by Study Visit (Week) in Adult Patients with MDD (Study 1)1
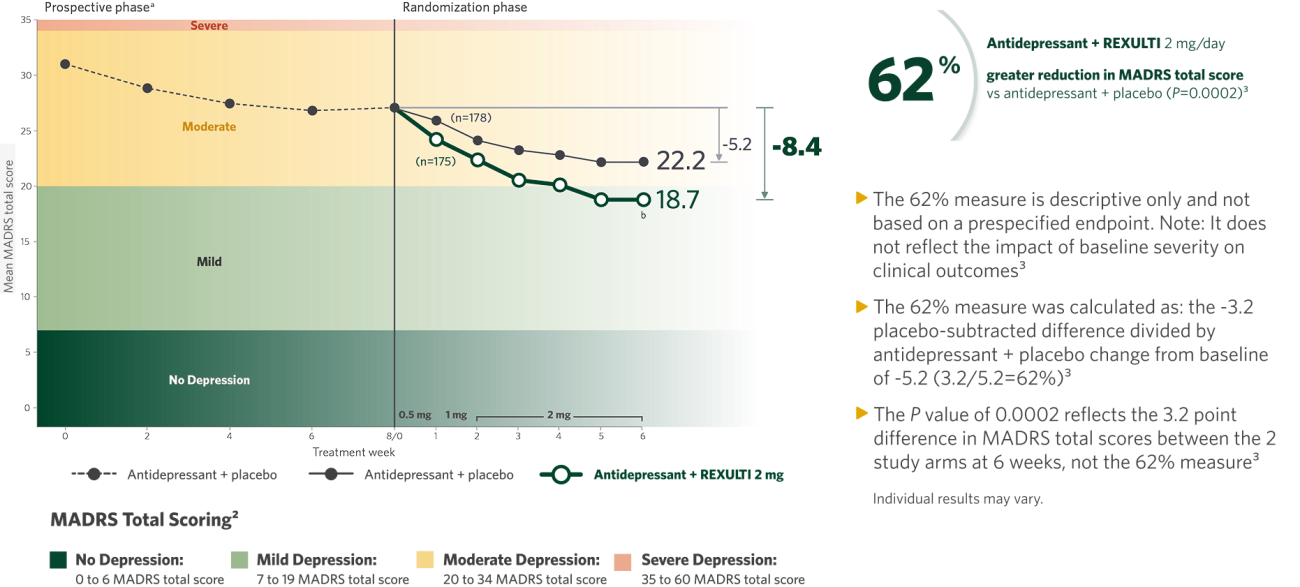
Mean Change from Baseline in MADRS Total Score by Study Visit (Week) in Adult Patients with MDD (Study 1)1
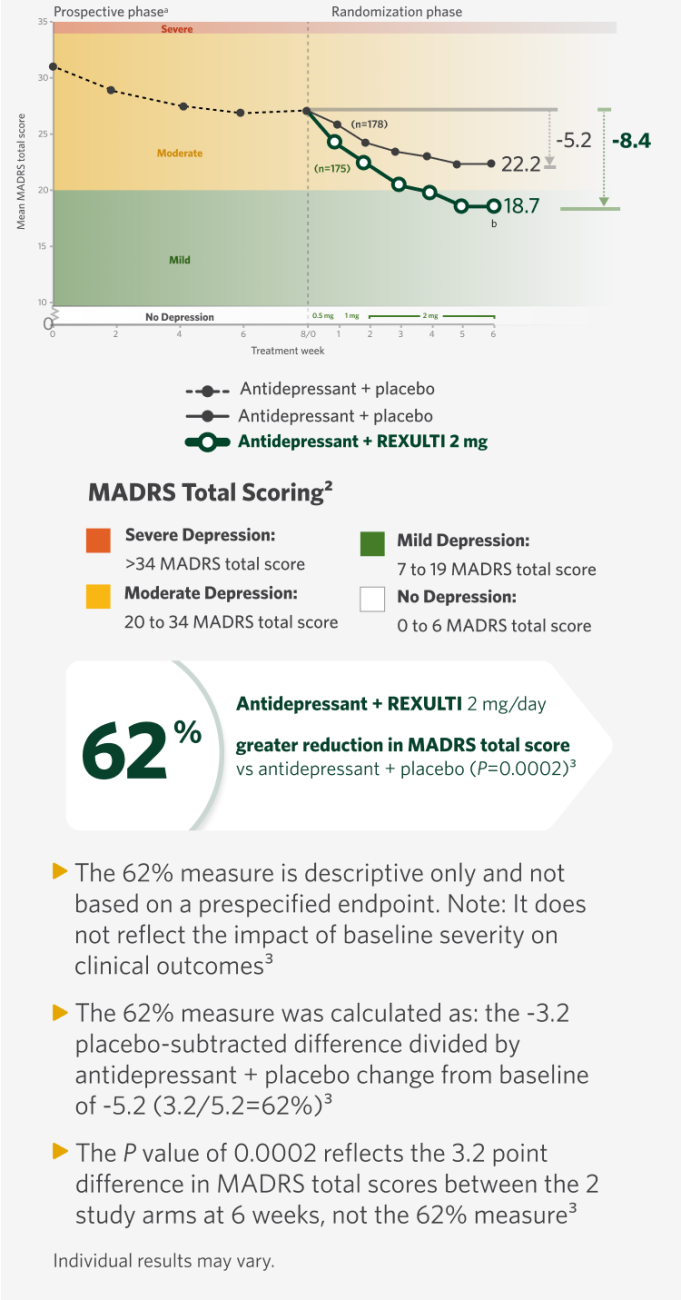
It is unknown if the differences observed at time points earlier than Week 6 represent clinically relevant treatment effects.1
aNot all patients had an inadequate response to antidepressant alone in the 8-week prospective phase. Only patients with an inadequate response entered the randomized double-blind treatment phase.
bP<0.001.
What is MADRS?
MADRS is a 10-item clinician-related scale used to assess the degree of depressive symptomatology, with 0 representing no symptoms and 60 representing worst symptoms
MADRS
clinician-rated
symptoms4:
-
- Apparent sadness
- Reported sadness
- Lassitude
- Inability to feel
-
- Inner tension
- Reduced appetite
- Reduced sleep
- Concentration difficulties
-
- Pessimistic thoughts
- Suicidal thoughts
Change in MADRS from Baseline at Week 6 (Primary Endpoint) in Adult Patients for Adjunctive Treatment of MDD (Studies 1 and 2)
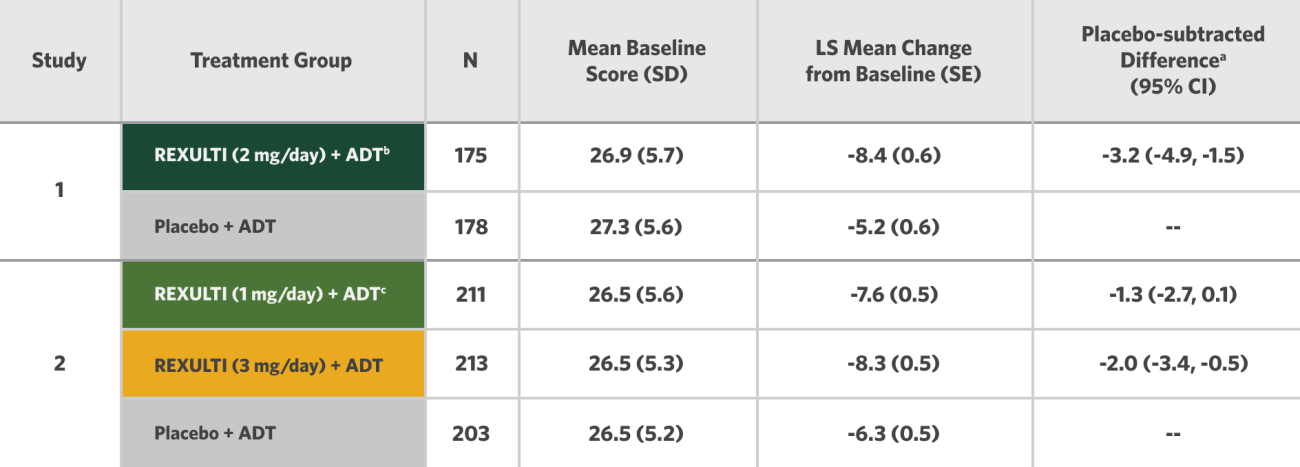
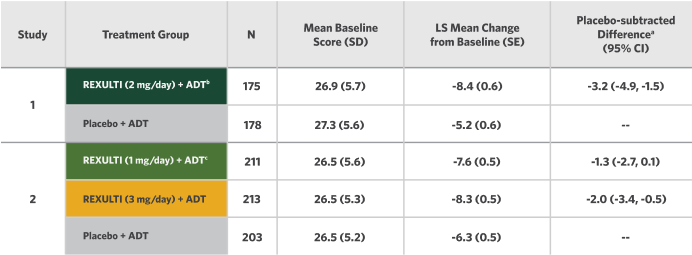
aDifference (drug minus placebo) in LS mean change from baseline.
bDosages statistically significantly superior to placebo.
cThe 1 mg/day dosage was studied in the trial, but is not recommended for the treatment of MDD.
ADT, antidepressant treatment; CI, unadjusted confidence interval; LS, least squares; MADRS, Montgomery-Åsberg Depression Rating Scale; MDD, major depressive disorder; SD, standard deviation; SE, standard error.
An examination of population subgroups did not suggest differential response based on age, gender, race, or choice of prospective antidepressant.
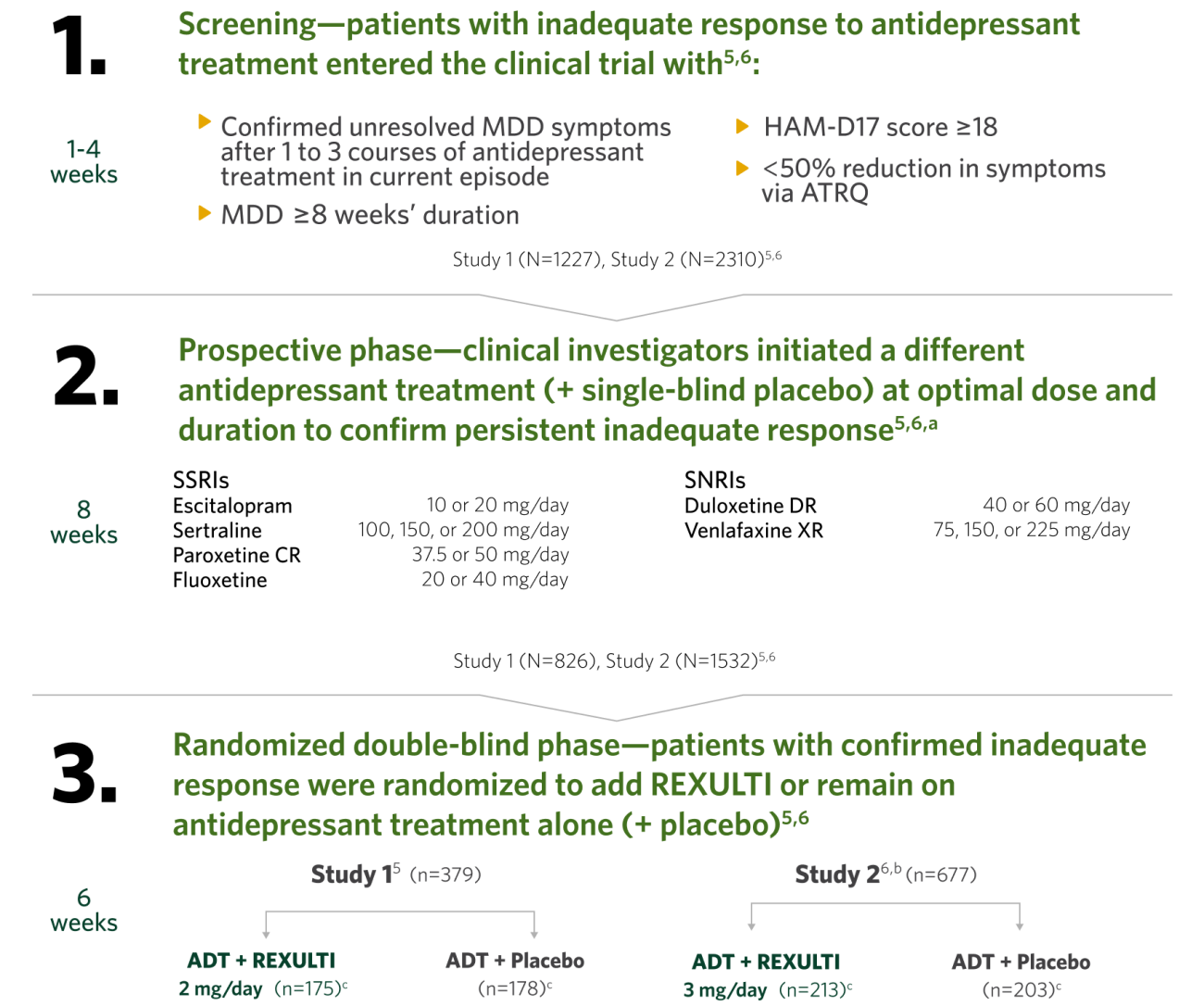
MDD study design and efficacy summary
The efficacy of REXULTI in the adjunctive treatment of major depressive disorder (MDD) was evaluated in two 6-week, double-blind, placebo-controlled, fixed-dose studies of adult patients meeting DSM-IV-TR criteria for MDD, with or without symptoms of anxiety, who had an inadequate response to prior antidepressant therapy (1 to 3 courses) in the current episode. After a screening phase of 1-4 weeks, patients entered into an 8-week prospective treatment phase with an SSRI or SNRI (+ single-blind placebo). Subsequently, patients having persistent symptoms without substantial improvement throughout the course of treatment and who met inclusion criteria were randomized to receive adjunctive REXULTI or placebo.5,6
In Study 2 for the REXULTI (3 mg/day) + ADT treatment group, the placebo-subtracted difference was -2.0 with a 95% CI (-3.4, -0.5).
Primary endpoint was the mean change from baseline to Week 6 in the MADRS total score in the randomization phase.5,6
The efficacy and safety of REXULTI were also studied in patients randomized to receive 1 mg/day in Study 2 (n=211). Results for the ADT + REXULTI 1 mg group for the primary efficacy parameter were not statistically significant when compared with ADT + placebo.6
ADT, antidepressant treatment; ATRQ, Antidepressant Treatment Response Questionnaire; CR, controlled release; DR, delayed release; DSM-IV-TR, Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (Text Revision); HAM-D17, 17-Item Hamilton Depression Rating Scale; MADRS, Montgomery-Åsberg Depression Rating Scale; MDD, major depressive disorder; SD, standard deviation; SE, standard error; SNRI, serotonin and norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor; XR, extended release.