Is partial response good enough?
Major depression among US adults has continued to increase1
Major depression among US adults has continued to increase1
Despite evidence supporting appropriate use, a chart review study showed that a patient may undergo ~5 treatment changes before being prescribed an adjunctive atypical antipsychotic5


Antidepressant treatment changes may affect response rates. In the STAR*D study, rates of response decreased with each additional switch in antidepressant treatment (48.6% in Step 1 to 16.3% in Step 4)3,b,d
Atypical antipsychotics may increase response rates for patients
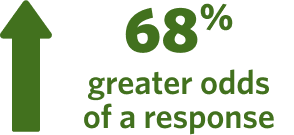

According to a meta-analysis, the chance of response doubled in patients treated with adjunctive atypical antipsychotics vs antidepressant treatments alone (odds ratio=1.68)6,e
aHave had one major depressive episode in the last year based on data from the 2023 National Survey on Drug Use and Health.2
bDemonstrated in ~3700 adults with MDD prescribed an antidepressant in the STAR*D study. Remission was defined as QIDS-SR16 ≤5 at 12-14 weeks; 36.8% achieved remission after initial treatment. Statistic extrapolated from STAR*D.2
cThe HAM-D has been the metric of choice for measuring depression in research settings. Other scales such as the MADRS and the Inventory for Depressive Symptomatology have also been used.4
dThe patient sample received successive acute treatment steps: 3671 patients entered at Step 1; 1439 patients continued at Step 2; 390 patients proceeded to Step 3; 123 patients advanced through all 4 steps. After SSRI monotherapy in Step 1, treatment options included switching medications or augmentation with either medication or cognitive therapy. Adjunctive atypical antipsychotics were not included at any step. Patients who either did not achieve response with a treatment or were unable to tolerate a treatment were encouraged to move to the next step.3
eIn a meta-analysis, response was defined as a 50% improvement from baseline to endpoint on either the MADRS or HAM-D17. Meta-analysis included 17 randomized trials with 3807 patients (duration range: 4-12 weeks) comparing adjunctive antipsychotic treatment to SSRI/SNRI treatment in adult patients (age range: 18-65 years) with MDD. There was a 68% greater odds of response from the antidepressant + adjunctive antipsychotic group vs the placebo + antidepressant group. Discontinuation due to adverse effects were 9.1% in the adjunctive antipsychotic treatment arm vs. 2.6% in the placebo arm.6
HAM-D17, 17-item Hamilton Depression Rating Scale; MADRS, Montgomery-Åsberg Depression Rating Scale; MDD, major depressive disorder; PHQ-9, Patient Health Questionnaire-9; QIDS-SR16, Quick Inventory of Depressive Symptomatology-Self Report 16; SNRI, serotonin and norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor; STAR*D, Sequenced Treatment Alternatives to Relieve Depression.
Watch Dr. Jain discuss the prevalence of partial response
in MDD and considerations for addressing it

Rakesh Jain, MD, MPH
Clinical Professor
Department of Psychiatry
Texas Tech University School of Medicine
Austin, Texas
The presenter is a paid consultant of Otsuka America Pharmaceutical, Inc. and Lundbeck.
Review treatment history data for patients with MDD experiencing partial response to antidepressants, and learn about REXULTI as an adjunctive treatment option.
